Isolation and Identification of Potent Antidiabetic Compounds from Antrodia cinnamomea-An Edible Taiwanese Mushroom
Abstract
Antrodia cinnamomea (AC), an edible Taiwanese mushroom, has been recognized as a valuable natural resource with vast biological and medicinal benefits. Recently, the hypoglycemic and anti-diabetic effects of AC were mentioned in several studies. However, no studies have investigated α-glucosidase inhibitors from AC fruiting bodies (ACFB) as they relate to type 2 diabetes (T2D) treatment. The purpose of this study was to gain evidence of potent α-glucosidase inhibitory effects, as well as isolate, identify and characterize the active compounds of ACFB. The MeOH extract of ACFB demonstrated potent α-glucosidase inhibitory activity, and possessed high pH stability (pH 2⁻11) and thermostable properties at 40⁻50 °C. Further purification led to the isolation of eight constituents from ACFB, identified as: 25S-antcin K (1), 25R-antcin K (2), dehydrosulphurenic acid (3), 25S-antcin I (4), 25S-antcin B (5), 25R-antcin B (6), dehydroeburicoic acid (7) and eburicoic acid (8). Notably, the ACFB extract and its identified compounds, except 1, 4, and 6 demonstrated a greater effect (EC50 = 0.025⁻0.21 mg/mL) than acarbose (EC50 = 0.278 mg/mL). As such, these active compounds were determined to be new potent mushroom α-glucosidase inhibitors. These active compounds were also identified on the HPLC fingerprints of ACFB.
Figures 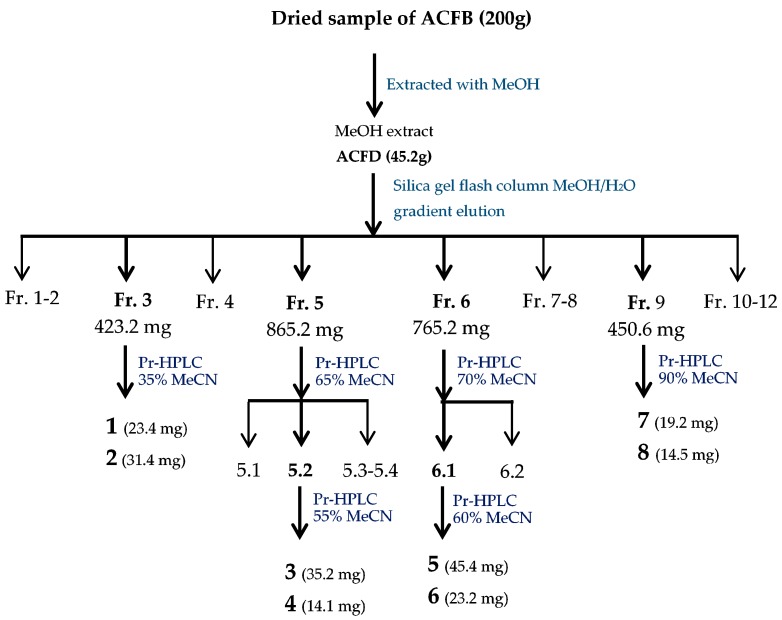
Figure 1
Flow chat of the purification…
Figure 1
Flow chat of the purification process of active compounds from Antrodia cinnamomea fruiting…
Figure 1 Flow chat of the purification process of active compounds from Antrodia cinnamomea fruiting bodies (ACFB) extract.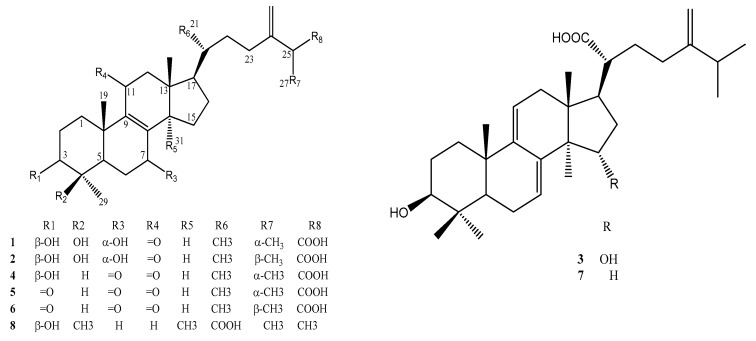
Figure 2
Chemical structures of purified active…
Figure 2
Chemical structures of purified active compounds. 25 S -antcin K ( 1 ),…
Figure 2 Chemical structures of purified active compounds. 25S-antcin K (1), 25R-antcin K (2), dehydrosulphurenic acid (3), 25S-antcin I (4), 25S-antcin B (5), 25R-antcin B (6), dehydroeburicoic acid (7) and eburicoic acid (8).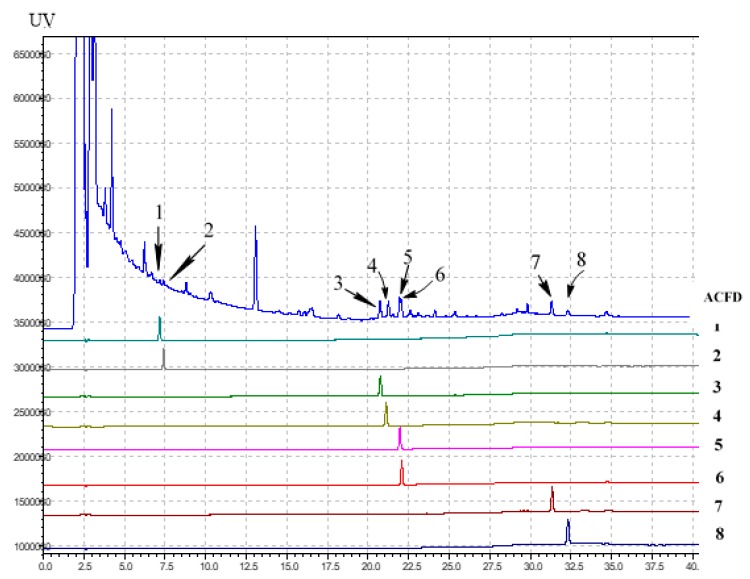
Figure 3
Identification of active inhibitors on…
Figure 3
Identification of active inhibitors on the HPLC fingerprints of ACFB extract. 25 S…
Figure 3 Identification of active inhibitors on the HPLC fingerprints of ACFB extract. 25S-antcin K (1), 25R-antcin K (2), dehydrosulphurenic acid (3), 25S-antcin I (4), 25S-antcin B (5), 25R-antcin B (6), dehydroeburicoic acid (7) and eburicoic acid (8). Analysis conditions: The mobile phase consisted of water containing 0.1% phosphoric acid and acetonitrile (ACN) using a gradient program of 40–50% ACN from 0−12 min, 50−60% ACN from 12−17 min, 60−95% ACN from 17−26 min and 95−100% ACN from 26−50 min; separation column (Cosmosil 5C18-AR-II, 5 μm, 250 × 4.6 mm i.d.) was employed, eluting at a flow rate of 1.0 mL/min at 35 °C; the real-time UV absorption was detected at 210 nm.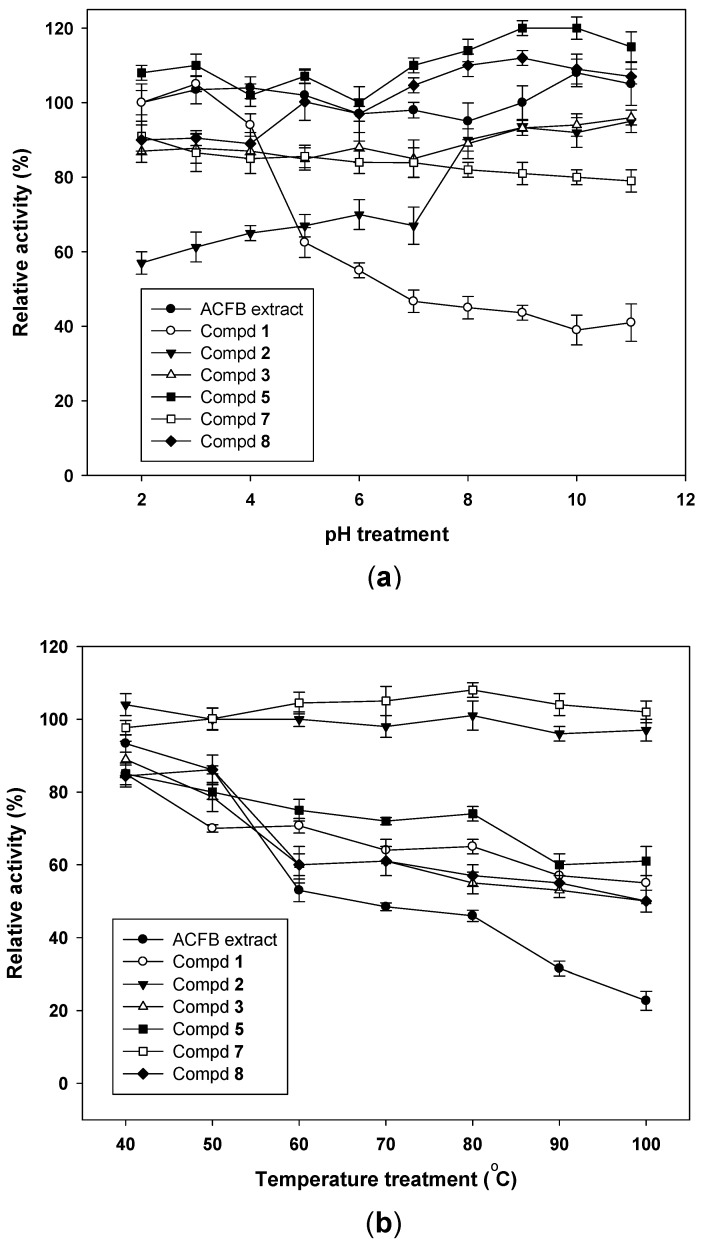
Figure 4
The pH and thermal stabilities…
Figure 4
The pH and thermal stabilities of ACFB extract and the purified compounds. The…
Figure 4 The pH and thermal stabilities of ACFB extract and the purified compounds. The pH (a) and thermal (b) stability of ACFB and its purified compounds were tested by treating the samples with a range of pH (2–11) and temperatures (40–100 °C) for 30 min, respectively. The α-glucosidase inhibition of treated samples was tested under the same conditions, using the bioassay mentioned in the methods section. Tests were performed in triplicate. Results are means ± SD.