Genome mining of cryptic bisabolenes that were biosynthesized by intramembrane terpene synthases from Antrodia cinnamomea
Abstract
Terpenoids represent the largest structural family of natural products (NPs) and have various applications in the pharmaceutical, food and fragrance industries. Their diverse scaffolds are generated via a multi-step cyclization cascade of linear isoprene substrates catalysed by terpene synthases (TPSs). Bisabolene NPs, which are sesquiterpenes (C15), have wide applications in medicines and biofuels and serve as bioactive substances in ecology. Despite the discovery of some canonical class I TPSs that synthesize bisabolenes from plants, bacteria and insects, it remained unknown whether any bisabolene synthases from fungi could produce bisabolenes as a main product. Antrodia cinnamomea, a Basidiomycota fungus, is a medicinal mushroom indigenous to Taiwan and a known prolific producer of bioactive terpenoids, but little is known regarding the enzymes involved in the biosynthetic pathways. Here, we applied a genome mining approach against A. cinnamomea and discovered two non-canonical UbiA-type TPSs that both synthesize (+)-(S,Z)-α-bisabolene (1). It was determined that two tailoring enzymes, a P450 monooxygenase and a methyltransferase, install a C14-methyl ester on the bisabolene scaffold. In addition, four new bisabolene derivatives, 2 and 4-6, were characterized from heterologous reconstitution in Saccharomyces cerevisiae. Our study uncovered enzymatic tools to generate structurally diverse bisabolene NPs. This article is part of the theme issue 'Reactivity and mechanism in chemical and synthetic biology'.
Figures 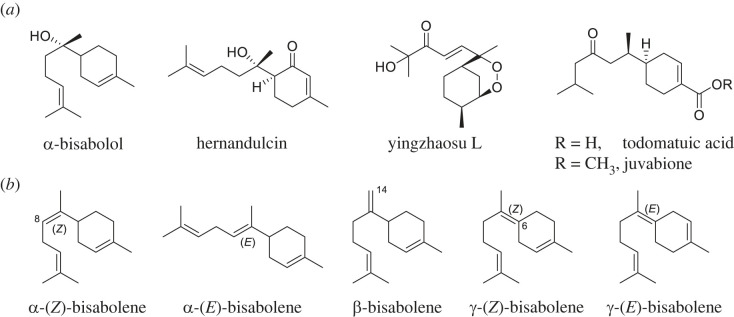
Figure 1.
( a ) NPs derived…
Figure 1.
( a ) NPs derived from the bisabolene scaffold. ( b ) Structure…
Figure 1. (a) NPs derived from the bisabolene scaffold. (b) Structure of bisabolene isomers.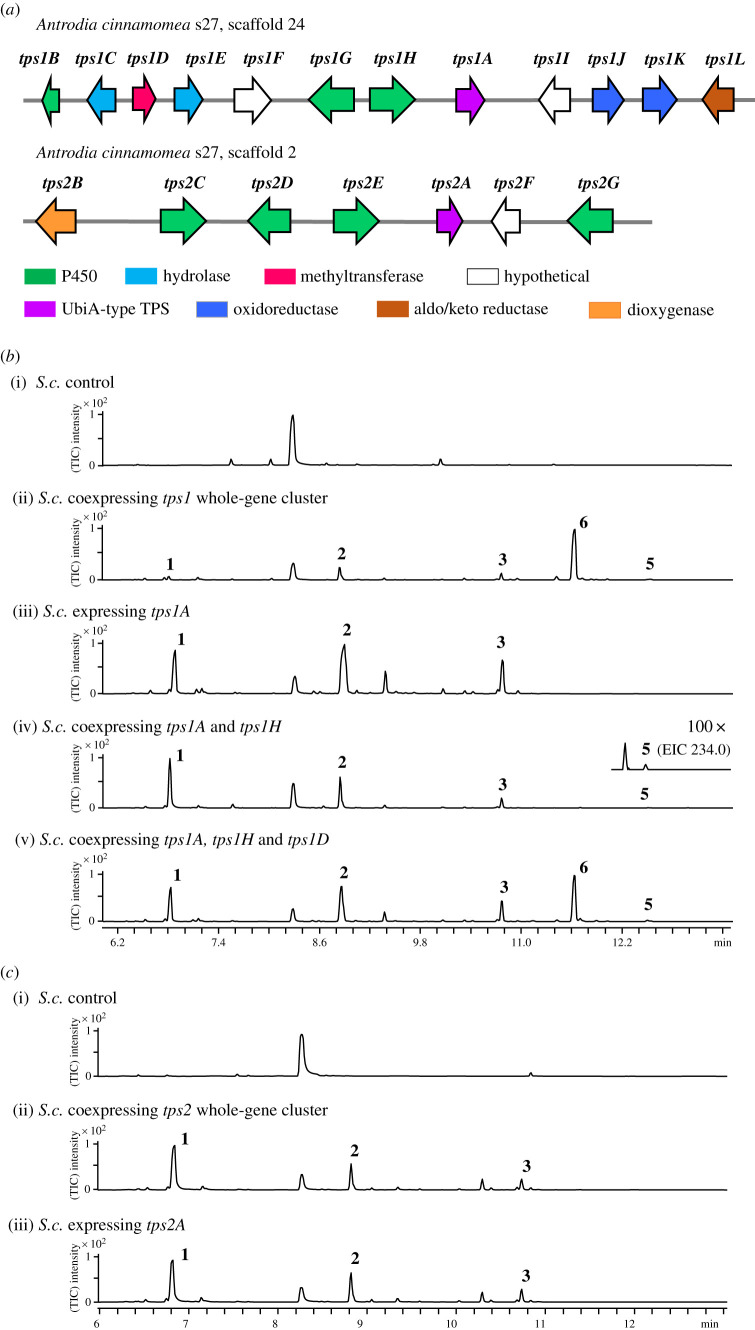
Figure 2.
Heterologous expression of tps1 and…
Figure 2.
Heterologous expression of tps1 and tps2 BGCs in S. cerevisiae cultured for 5…
Figure 2. Heterologous expression of tps1 and tps2 BGCs in S. cerevisiae cultured for 5 days. (a) BGCs of tps1 and tps2 in A. cinnamomea; (b) SPME-GC-MS analysis of compounds produced through different combinations of tps1 and (c) tps2 genes. (Online version in colour.)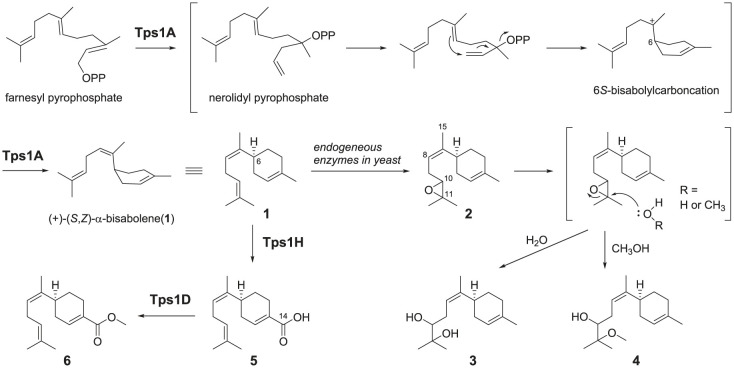
Scheme 1.
Proposed biosynthetic pathway to 6…
Scheme 1.
Proposed biosynthetic pathway to 6 through the combination of tps1A , tps1H and…
Scheme 1. Proposed biosynthetic pathway to 6 through the combination of tps1A, tps1H and tps1D of the tps1 BGC from A. cinnamomea.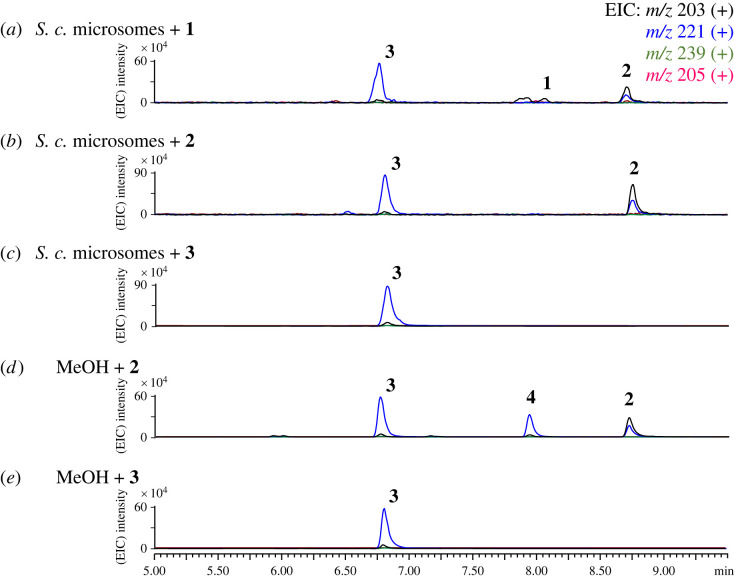
Figure 3.
LC-MS analysis of the in…
Figure 3.
LC-MS analysis of the in vitro reaction of the microsome fractions of S.…
Figure 3. LC-MS analysis of the in vitro reaction of the microsome fractions of S. cerevisiae with (a) 1, (b) 2 and (c) 3. (d) Incubation of methanol with 2 and (e) 3 for 16 h. EIC, elective ionization. (Online version in colour.)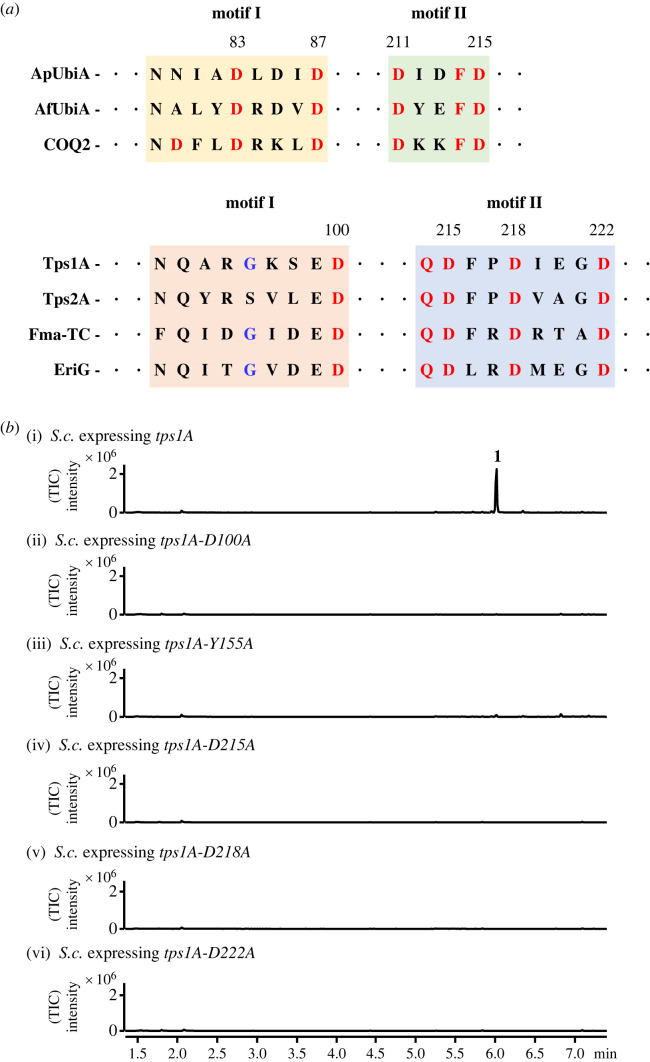
Figure 4.
Verification of Tps1A active site…
Figure 4.
Verification of Tps1A active site motifs. ( a ) Alignment of amino acid…
Figure 4. Verification of Tps1A active site motifs. (a) Alignment of amino acid sequences of the two conserved aspartate-rich motifs of the UbiA prenyltransferase family and the motifs of UbiA-like TPSs. (b) SPME-GC-MS analysis of yeast expressing the tps1A mutants D100A, D215A, D218A, D222A and Y155A. TIC, total ion content. (Online version in colour.)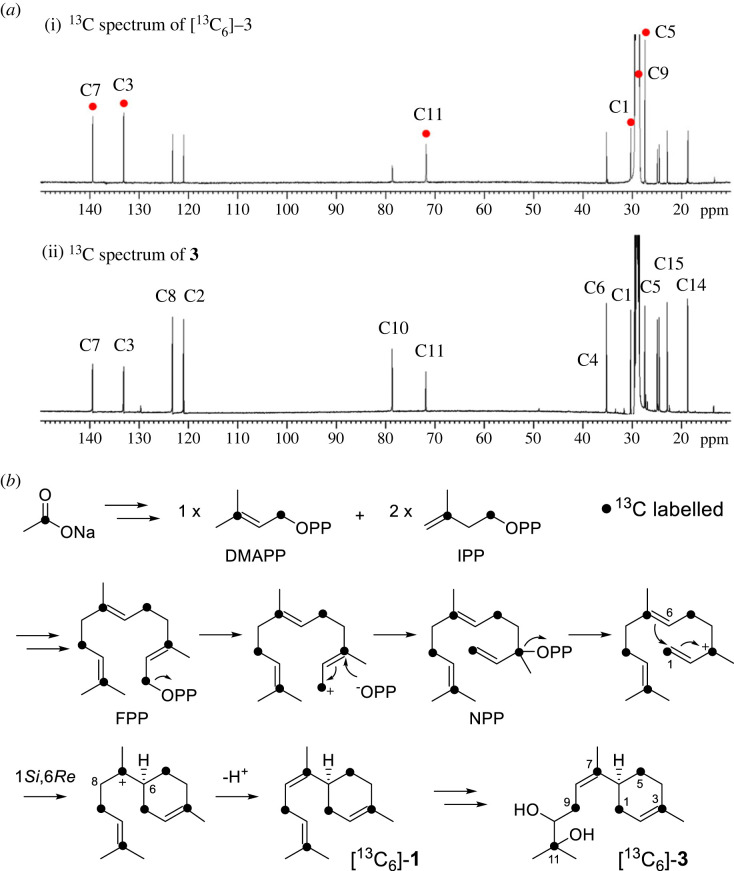
Figure 5.
The labelling experiments with […
Figure 5.
The labelling experiments with [ 13 C 6 ]-3 using 1- 13 C-sodium…
Figure 5. The labelling experiments with [13C6]-3 using 1-13C-sodium acetate. (a) The comparison of the 13C-NMR spectra of (i) 13C-labelled and (ii) unlabelled 15. (b) Proposed mechanism of the cyclization of FPP to 1 and generation of 3. (Online version in colour.)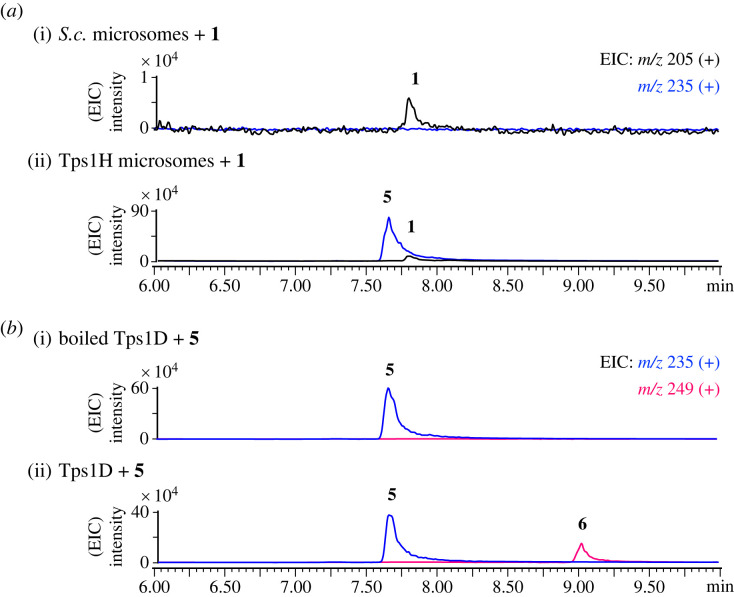
Figure 6.
Verification of the function of…
Figure 6.
Verification of the function of Tps1H and Tps1D. LC-MS profiles of in vitro…
Figure 6. Verification of the function of Tps1H and Tps1D. LC-MS profiles of in vitro reactions of (a) Tps1H for the conversion of 1 to 5 and (b) Tps1D for the conversion of 5 to 6.(Online version in colour.)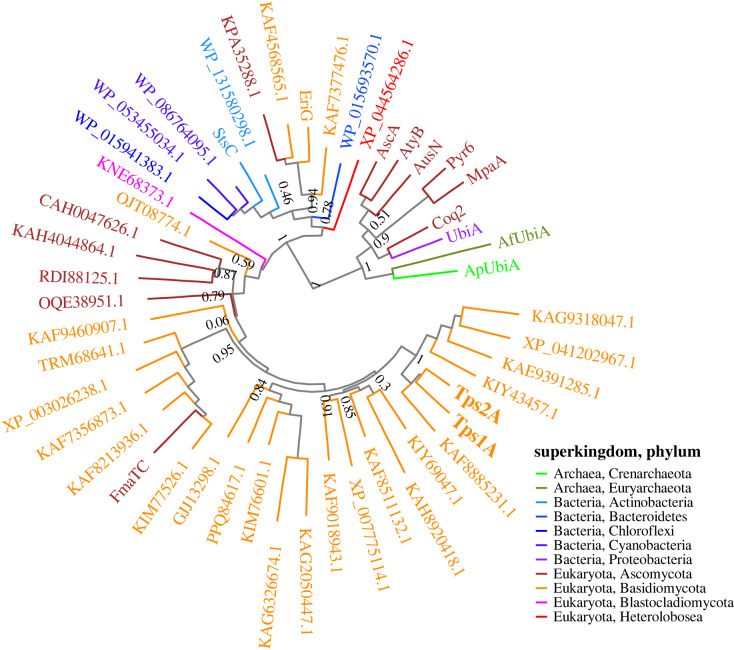
Figure 7.
Phylogenetic analysis of Tps1A and…
Figure 7.
Phylogenetic analysis of Tps1A and Tps2A bisabolene synthases and UbiA-type TPSs, the UbiA…
Figure 7. Phylogenetic analysis of Tps1A and Tps2A bisabolene synthases and UbiA-type TPSs, the UbiA superfamily of intramembrane prenyltransferases and integral membrane TPSs from different organisms.(Online version in colour.) All figures (8)